RFID for Healthcare
RFID empowers hospitals, medical device manufacturers, and pharma to track and digitize critical inventory
Hospitals, medical device manufacturers, and pharmaceutical companies rely on RFID technology to automate workflows, streamline inventory management, and ensure real-time tracking—enhancing operational efficiency, patient safety, and regulatory compliance across the healthcare sector.
Xerafy pioneered autoclavable RFID tags that maintain reliable performance in the most demanding sterilization environments. Our rugged solutions, co-developed with industry leaders, meet the highest standards of the healthcare sector. These tags and labels have been validated through diverse use cases, solving critical challenges in sterile processing, medical supplies replenishment, pharmacy inventory management, and textile tracking.
Xerafy RFID for Healthcare capabilities are showcased in detailed case studies, demonstrating the measurable benefits of RFID tracking in real-world environments.
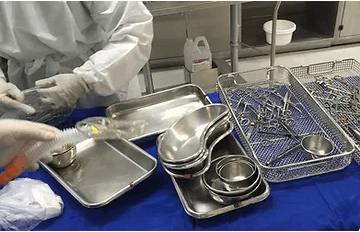
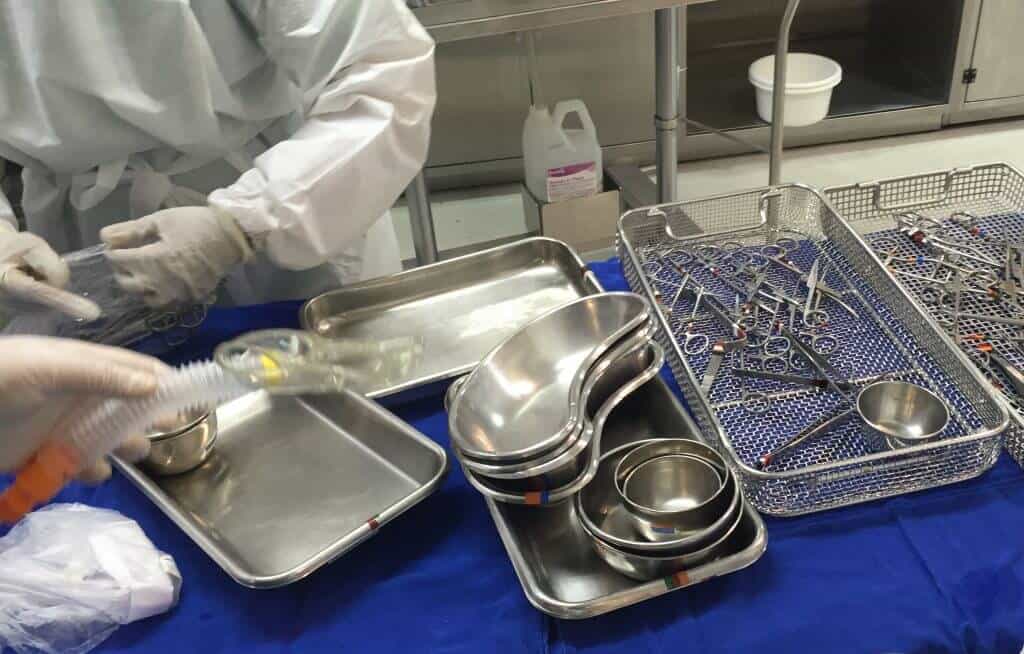
Sterile Processing Departments (SPDs) play a crucial role in cleaning, disinfecting, sterilizing and assembling devices and instruments for the surgical team. RFID technology transforms sterile processing workflows by providing precise tracking and control over these workflows.
A hospital SPD faces numerous challenges in managing surgical trays and ensuring efficient sterile processing:
Inefficient Reprocessing Workflows: Manual tracking during sterilization and reprocessing introduces errors and delays, hindering surgical readiness and SPD efficiency.
Inventory Visibility Issues: Limited real-time visibility of surgical trays and instruments can lead to misplaced items and last-minute procedural disruptions.
Compliance Requirements: Hospitals must follow strict sterilization protocols and maintain precise records to meet regulatory and audit requirements.
Operational Strain: SPD staff and perioperative teams face increased workloads when manual processes fail to ensure timely tray availability, creating bottlenecks in critical workflows.
Xerafy’s autoclavable RFID tags provide a robust solution for surgical tray tracking and sterile processing by seamlessly integrating with hospital inventory and sterilization management systems:
Accurate Tracking During Reprocessing: RFID tags attached to surgical trays and instruments allow for automated and precise tracking throughout the sterilization cycle.
Real-Time Inventory Management: RFID technology offers real-time visibility into the location and status of surgical assets, ensuring timely availability for procedures.
Rugged Autoclavable Tags: Xerafy’s tags are designed to withstand high temperatures and harsh sterilization conditions, ensuring consistent performance.
Streamlined Workflows: Automated RFID tracking lightens the workload for SPD personnel and perioperative teams, enabling them to focus on essential tasks.
Compliance Support: RFID-enabled systems simplify record-keeping, ensure adherence to sterilization and regulatory standards, and provide compatibility with leading hospital management platforms.
Sterile processing tracking systems implemented with Xerafy’s RFID solutions deliver measurable advantages:
Enhanced Efficiency: Streamlined sterilization workflows reduce delays and improve surgical readiness.
Improved Asset Utilization: Real-time tracking minimizes loss and ensures optimal use of surgical trays and instruments.
Regulatory Assurance: Automated data capture ensures compliance with sterilization protocols and audit requirements.
Reduced Operational Costs: Eliminating inefficiencies and errors leads to cost savings across sterile processing workflows.
Xerafy’s RFID technology enables hospital SPDs to manage sterile inventory with precision, efficiency, and compliance, enhancing hospital operations and infection prevention.
Loaner systems temporarily provide critical instruments and implants for healthcare procedures. Managed by medical device manufacturers, distributors, and institutions, these systems rely on RFID technology to manage consignment inventory and track loaner devices effectively. This ensures the availability of surgical sets, implants, and devices critical to patient care.

Effective management of loaners and consigned inventory directly impacts an institution’s operational efficiency, patient safety, and regulatory compliance:
Traceability Issues: Manual tracking methods complicate tracing loaner devices, disrupting workflows such as surgical set preparation, inventory audits, and equipment allocation, thereby increasing the risk of errors.
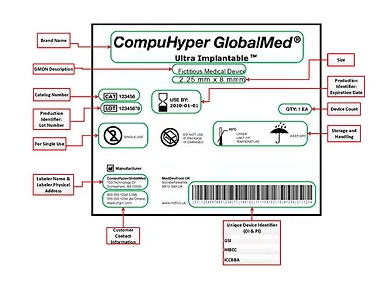
Regulatory Compliance: Healthcare regulators, including the FDA, mandate UDI traceability for medical device inventory. RFID technology supports compliance by automating data capture and ensuring traceability across the supply chain.
Loss and Misplacement: High-value equipment is at risk of being lost or delayed during transit or at healthcare facilities.
Inefficient Reconciliation Processes: Manual reconciliation of surgical sets and implants after procedures can be time-consuming and prone to errors.
Xerafy’s rugged autoclavable RFID tags provide a healthcare-specific solution by offering unmatched durability in harsh healthcare environments:
UDI-Compliant Tracking: Embedded RFID tags provide precise and automated tracking of loaner devices, ensuring compliance with UDI mandates and global regulatory standards.
Real-Time Inventory Management: RFID-enabled containers and trays allow for real-time visibility of surgical sets and implants, whether in the field, in transit, or at healthcare facilities.
Sterilization-Ready Tags: Xerafy’s rugged autoclavable RFID tags are specifically designed to withstand repeated sterilization processes and harsh handling conditions, ensuring consistent performance and durability.
Automated Reconciliation: RFID technology streamlines post-procedure reconciliation, reducing manual effort and improving accuracy.
Data-Driven Insights: RFID systems enable manufacturers to gather anonymous usage data, improve recall programs, and enhance procurement processes.
Xerafy’s autoclavable RFID tagging solutions address the key challenges of loaner system management by:
Automating Traceability: Accurately track devices across their lifecycle.
Ensuring Compliance: Automate UDI data capture to meet regulatory standards.
Improving Efficiency: Streamline inventory management and post-procedure reconciliation.
Reducing Costs: Prevent loss and unnecessary purchases of high-value devices.
Hospital equipment such as wheelchairs, IV pumps, and portable diagnostic tools are essential at the point of care. Ensuring their availability and functionality impacts operational efficiency, patient outcomes, and staff productivity in a healthcare environment.
![]()
Hospitals often struggle to locate critical equipment, leading to:
Delays in Patient Care: Nurses spend significant time searching for items like wheelchairs or infusion pumps, which can lead to delays in treatments or procedures.
Surgical Delays: Essential equipment may be misplaced or unavailable when needed for surgeries, risking patient safety and operational schedules.
Costly Redundancies: To compensate for lost or misplaced equipment, hospitals frequently make unnecessary purchases, increasing operational costs.
Manual Tracking Inefficiencies: Barcode-based systems require line-of-sight and manual scanning, which are time-consuming and prone to errors.
Xerafy’s RFID tagging solutions provide a cutting-edge approach to hospital equipment tracking by offering advanced durability, seamless integration, and cost-effective automation that set them apart:
Real-Time Location Tracking: Utilizing passive UHF RFID technology and Real-Time Location Systems (RTLS), Xerafy enables hospitals to pinpoint the last-known or real-time location of equipment across facilities.
Contactless and Automated Operation: RFID tags eliminate the need for line-of-sight scanning, automating the tracking process and significantly reducing manual effort.
Durable Tags and Labels: Designed to withstand hospital environments, Xerafy’s tags are built to endure washing processes and resist physical wear. Unlike traditional barcode systems or less durable RFID options, they maintain reliable performance even in demanding conditions, ensuring effective tracking of mobile equipment.
Seamless Integration: Xerafy’s RFID tags integrate effortlessly with existing hospital management systems, offering a scalable and field-ready solution.
Implementing Xerafy’s RFID tagging systems deliver measurable outcomes:
Time Savings: Staff spend less time locating equipment, enabling them to focus on patient care.
Improved Operational Efficiency: Equipment availability and utilization rates improve, preventing delays in treatments or procedures.
Cost Reduction: Avoid unnecessary equipment purchases by ensuring existing assets are fully utilized.
Enhanced Compliance and Safety: Automated tracking supports audit readiness and compliance with healthcare regulations.
Xerafy’s RFID hospital equipment tagging solutions optimize workflows, ensuring critical equipment is available when needed.
Effective pharmacy inventory management is critical for preventing shortages, managing controlled substances, and ensuring compliance with strict regulatory requirements. RFID technology automates these processes, providing real-time insights and reinforces accountability.

Managing pharmacy inventory requires addressing highly specialized challenges unique to medication handling and distribution:
Controlled Substance Oversight: Pharmacies must securely manage narcotics and other controlled substances with detailed audit trails to meet legal requirements.
Complex Prescription Fulfillment: Managing high volumes of prescriptions alongside manual inventory checks increases the risk of errors and delays.
Critical Expiry and Recall Management: Pharmacies must ensure medications nearing expiration are flagged in real time and recalled drugs are swiftly removed to protect patient safety.
Demand Forecasting and Stock Allocation: Without accurate data, pharmacies struggle to predict medication demand and allocate stock efficiently across departments.
Xerafy’s RFID tagging technology simplifies pharmacy inventory management by:
Real-Time Medication Monitoring: RFID tracks every vial and blister pack in real time, ensuring accurate stock levels and preventing critical shortages.
Secure Controlled Substance Management: RFID ensures secure tracking and handling of controlled substances, supporting regulatory compliance.
Expiry and Recall Management: RFID automates the tracking of expiry dates and facilitates swift identification of affected items during recalls.
Integrated Dispensing Accuracy: RFID ensures medications are dispensed correctly and securely, reducing human error in high-volume environments.
Implementing Xerafy’s RFID tagging solutions delivers measurable outcomes:
Enhanced Patient Safety: Ensures the availability of safe and effective medications through precise inventory management.
Improved Compliance: Simplifies adherence to regulatory standards for controlled substances and medication tracking.
Pharmacy Workflow Optimization: Automates routine inventory tasks, freeing pharmacists to focus on patient counseling and clinical activities.
Cost Savings: Minimizes waste and optimizes stock levels through real-time insights.
Hospitals rely on efficient inventory workflows to ensure timely replenishment of storerooms, track expiry dates, and manage recalls, all critical to patient safety and operations. RFID technology automates these processes, reducing errors and improving accuracy.

Hospitals face key challenges in managing medical supplies effectively:
Traceability and Compliance: Tracking expiry dates, managing recalls, and maintaining UDI-compliant traceability are essential for ensuring patient safety and operational integrity.
Inefficient Replenishment Processes: Manual processes cause restocking delays and inaccuracies, impacting supply availability in the warehouse and storerooms.
Complex Inventory Systems: Hospitals must efficiently manage diverse inventories, such as consumables and consigned goods, to ensure availability and compliance.
Data Gaps: Lack of real-time visibility into stock levels and consumption patterns hinders effective inventory planning.
Xerafy’s RFID tracking technology simplifies inventory management by:
Real-Time Inventory Visibility: RFID enables hospitals to monitor storeroom stock levels, ensure timely replenishment, and automate new orders.
Enhanced Traceability and Compliance: RFID tags enable hospitals to track expiry dates, quickly locate affected items during recalls, and maintain UDI-compliant traceability, ensuring patient safety and compliance.
Streamlined Storeroom Inventory: RFID integrates with IoT shelf sensors, Kanban bins, and dispensing machines to improve real-time visibility and automate inventory management.
Optimized Consigned Inventory Management: RFID ensures transparent billing, tracks deliveries, and supervises consigned inventory for greater accountability.
Implementing Xerafy’s RFID tagging solutions delivers measurable outcomes:
Improved Patient Safety: Accurate traceability and secure distribution ensure the right products reach patients safely.
Operational Efficiency: RFID automation makes inventory access and stocking more effective and efficient.
Cost Savings: Real-time tracking optimizes storeroom inventory levels, minimizing waste and reducing unnecessary procurement.
Regulatory Compliance: Automated data capture supports adherence to UDI and other regulatory requirements.
Xerafy’s RFID technology helps hospitals manage supplies with precision and efficiency, improving operations and patient outcomes.
Hospitals rely on a consistent supply of scrubs, linens, gowns, and other textiles to maintain hygiene standards, ensure patient comfort, and support clinical workflows. Managing these high-turnover items efficiently is essential to prevent shortages, reduce waste, and meet infection control standards. RFID technology provides a robust solution for tracking and managing textile inventory, offering real-time insights and automation.
Managing hospital textile inventory involves unique operational challenges:
High Turnover and Loss Rates: Frequent use, laundering, and misplacement of textiles result in high replacement costs and operational inefficiencies.
Hygiene and Infection Control: Ensuring that clean and sterile linens and gowns are always available is critical for infection prevention and patient safety.
Inefficient Tracking: Manual methods of tracking textile usage and laundry cycles are time-consuming and prone to errors, leading to inventory shortages.
Cost Management: Overstocking textiles increases costs, while understocking risks disruptions in patient care and clinical operations.
Xerafy’s RFID textile inventory tracking technology streamlines hospital laundry operations by:
Automating Laundry and Usage Tracking: RFID tags embedded in textiles allow hospitals to monitor laundry cycles, track usage, and optimize inventory turnover.
Ensuring Hygiene Compliance: RFID enables hospitals to verify that textiles meet hygiene standards, ensuring only sterilized items are available for use.
Real-Time Inventory Visibility: RFID provides real-time insights into textile availability, helping staff quickly locate items and avoid shortages.
Loss Prevention: RFID tracks textiles throughout their lifecycle, reducing losses and enabling accountability for misplaced or stolen items.
Optimizing Stock Levels and Sustainability: RFID data supports just-in-time replenishment, reducing waste, lowering operational costs, and contributing to sustainability efforts by extending textile lifecycles and minimizing overproduction.
Implementing Xerafy’s RFID textile tagging solutions delivers measurable benefits:
Improved Efficiency: Streamlines laundry operations and ensures textiles are always available where needed.
Enhanced Hygiene and Safety: Ensures compliance with infection control standards through verified tracking.
Cost Savings and Environmental Impact: Reduces replacement costs and waste through efficient inventory management, supporting greener hospital operations.
Accountability: Provides detailed audit trails to prevent loss and ensure proper usage of hospital textiles.
Case Studies RFID for Healthcare
From surgical trays to pharmacy inventory and textiles, see how Xerafy’s expertise and field-tested RFID technology are transforming healthcare asset and inventory management worldwide
Benefits of RFID For Healthcare
Real-Time Tracking
RFID enables precise tracking of medical equipment, supplies, and devices across healthcare settings, reducing losses and ensuring accurate location data in real-time or at the last-known location.
Operational Efficiency
RFID streamlines workflows by automating the tracking of items and assets, saving time for medical staff and reducing the risk of delays in procedures, patient care, or inventory replenishment.
Enhanced Automation
Unlike barcode-based systems, RFID provides contactless tracking at both the item and bulk level, supporting automated inventory management and operational processes in hospitals, clinics, pharmacies, and manufacturing environments.
Optimized Asset Management
With RFID, healthcare providers gain full visibility of assets, helping avoid unnecessary purchases, improving asset utilization, and ensuring that equipment is always available when needed.
Data-Driven Insights
RFID technology allows healthcare organizations to track inventory usage patterns and optimize stock levels. This data helps identify shortages, manage procurement cycles more efficiently, and improve decision-making for resource allocation.
Frequently Asked Questions about RFID for Healthcare
What types of healthcare assets and inventory can be tracked with RFID?
RFID technology provides real-time visibility and traceability across the healthcare ecosystem — from manufacturers and distributors to hospitals and pharmacies. Assets and inventory that can be tracked include:
+ Surgical trays and instruments – supporting sterile processing, validation of sterility, and cycle count automation.
+ Consignment and loaner kits – providing location and usage data across field kitting locations, medical device sales reps, and hospitals.
+ Point-of-care equipment – ensuring availability and reducing loss or misplacement.
+ Pharmaceuticals and vaccines – enabling cold chain monitoring with temperature logging to protect efficacy.
+ Hospital scrubs, gowns, and linens – improving turnaround and reducing shrinkage.
+ Lab equipment, samples, and consumables – enhancing accuracy, compliance, and operational efficiency.
+ Patients – with RFID-enabled wristbands for accurate identification and care delivery.
By covering this wide scope, RFID helps streamline operations, reduce inefficiencies, and ensure critical assets are always available when and where they are needed.
What are the challenges for RFID and wireless technologies in hospitals?
Healthcare environments are some of the most complex settings for wireless technologies. RFID systems must overcome unique challenges to deliver reliable performance:
Patient Safety
RFID is safe at the point of care. Regulatory authorities such as the FDA note that they are not aware of adverse events associated with RFID use in healthcare.
Sterilization and Autoclave
Surgical trays, instruments, and medical devices must endure repeated sterilization cycles (autoclave, EtO, gamma, e-beam). RFID tags must be specifically engineered to survive these harsh conditions without performance loss.
Integration and Infrastructure
Hospitals face strict regulatory approvals, limited downtime, and highly sensitive environments. RFID must be compact, non-disruptive, and interoperable with existing IT platforms while minimizing new infrastructure requirements.
Signal Interference
Hospitals contain dense metal structures, surgical trays, and equipment that can interfere with wireless signals. RFID solutions must be optimized for on-metal performance to ensure reliable reads in these conditions.
By addressing these challenges, RFID enables hospitals and medical device companies to deploy systems that are not only reliable but also safe, compliant, and seamlessly integrated into daily operations.
Can RFID tags survive autoclave and sterilization?
Yes. RFID tags designed for healthcare must withstand the same sterilization processes as the surgical trays and instruments they are attached to. Xerafy pioneered autoclavable RFID tags and continues to lead in this area:
+ Autoclave Cycles – Endure repeated steam sterilization at high temperatures and pressures.
+ Chemical Sterilants – Resistant to EtO and harsh cleaning agents used in sterile processing.
+ Radiation Sterilization – Tolerant to gamma and e-beam irradiation for medical device packaging.
+ Regulatory Alignment – Tested for biocompatibility and validated to support FDA and EU MDR compliance in sterile processing.
By surviving through these extreme conditions without performance loss, RFID sterile processing tracking systems ensure that instrument-level traceability and sterility validation are possible across the full lifecycle of medical devices.
How can RFID improve patient safety?
Patient safety is the driving force behind every RFID healthcare application. By providing real-time visibility and traceability, RFID helps reduce risks and ensure the right assets are in the right place at the right time:
+ Surgical Instruments and Trays – Verify that sterilized and validated sets are used, reducing the risk of errors in the operating room.
+ Unique Device Identification (UDI) – Ensure the correct device is selected for each procedure, supporting regulatory compliance and traceability.
+ Medication Accuracy – Prevent medication errors by linking RFID labeled drugs to patient records at the point of care.
+ Patient Identification – RFID wristbands connect patients to their medical records, enabling accurate treatment and reducing misidentification risks.
By embedding safety checks directly into workflows, RFID enables clinicians to focus more time on patient care and less on manual verification.
How does RFID improve compliance with FDA UDI and global regulations?
RFID enables automated, end-to-end traceability of medical devices, helping manufacturers and healthcare providers meet global identification and reporting requirements:
+ FDA Unique Device Identification (UDI) – Store and capture UDI data directly in the RFID tag’s memory, ensuring automatic identification at every stage of the device lifecycle.
+ EU Medical Device Regulation (MDR) – Support European compliance requirements by validating sterility, device use, and chain of custody.
+ Regulatory and Quality Validation – RFID tags can be tested in autoclaves and sterilization environments to meet strict quality and biocompatibility standards.
+ Global Standardization – Beyond FDA and EU MDR, RFID supports international traceability systems, helping organizations maintain compliance across multiple regions.
By embedding regulatory compliance into daily workflows, RFID reduces the burden of manual reporting and ensures that hospitals and manufacturers can demonstrate adherence to patient safety and traceability standards.
How does RFID improve operational efficiency across healthcare?
RFID automates the tracking and management of assets, reducing manual tasks and ensuring accurate, real-time data is available across the healthcare supply chain:
+ Hospitals – Automate inventory counts, streamline replenishment, and reduce delays in surgery by ensuring instruments and equipment are available when needed.
+ Field and Consignment – Track surgical trays and loaner kits across sales reps, trunk inventory, and hospital deliveries, minimizing loss and improving utilization.
+ Cycle Counts – Replace manual processes with automated RFID scans, enabling faster, more accurate audits with less disruption to operations.
+ Data-Driven Decisions – Provide analytics on usage patterns, location history, and inventory turnover, helping organizations optimize resources and reduce costs.
By creating a single, accurate source of truth for assets and inventory, RFID reduces inefficiencies and frees up clinical staff to spend more time on patient care.
How does RFID track consignment and loaner surgical kits?
Loaner and consignment systems are among the most complex inventory challenges in healthcare. RFID provides real-time visibility across the entire lifecycle of surgical kits, from manufacturer to patient use:
+ Field and Trunk Inventory – Track kits in sales reps’ vehicles and regional kitting centers, reducing lost sets and improving utilization.
+ Hospital Delivery and Return – Monitor the location and condition of kits as they move in and out of hospitals, ensuring timely availability for procedures.
+ Usage and Sterility Validation – Combine location data with temperature logging to validate sterility and record when consignment inventory has been used.
+ System Integration – Connect to existing loaner management systems, enabling seamless data exchange and automated cycle counts.
By automating these workflows, RFID reduces inefficiencies, improves kit utilization, and ensures that critical instruments are ready and compliant when needed.
What data and system integrations are available with RFID?
For RFID to deliver value in healthcare, it must integrate seamlessly into existing IT environments and provide transparent access to data. Key capabilities include:
+ System Integration – RFID platforms can connect with ERP, inventory, and loaner management systems such as SAP, Steris, Censis, as well as hospital EHR/EMR systems.
+ APIs and Data Exchange – Standard APIs allow real-time data flow between RFID readers, cloud platforms, and existing software, minimizing manual entry and duplication.
+ Analytics and Dashboards – RFID portals and dashboards provide visibility into inventory levels, usage patterns, and equipment status for decision-making and compliance.
+ Data Ownership and Export – Hospitals and manufacturers retain ownership of their RFID data, with the ability to export in raw or structured formats to meet reporting, regulatory, and operational requirements.
By ensuring interoperability, RFID solutions deliver accurate, actionable data where it is needed — without disrupting established workflows or IT infrastructure.
How does RFID support lab and clinical environments?
RFID provides accurate, automated tracking for assets, consumables, and samples across laboratory and clinical settings:
+ Equipment and Devices – Monitor the location, availability, and maintenance status of lab instruments and clinical devices, reducing downtime and loss.
+ Samples and Specimens – Assign unique identifiers to ensure correct matching with patient records, preserve chain of custody, and minimize handling errors.
+ Consumables – Automate the tracking of reagents, test kits, and supplies to ensure availability and reduce expiration-related waste.
+ Integration – Connect with laboratory information management systems (LIMS) and hospital IT platforms, enabling compliance reporting and real-time data access.
By digitizing workflows, RFID enhances accuracy, compliance, and efficiency in both labs and clinical environments.
What benefits does RFID provide for pharmacy and medication tracking, including vaccines and cold chain?
RFID strengthens safety, compliance, and efficiency in pharmaceutical management by providing end-to-end visibility from manufacturer to patient:
+ Medication Safety – Verify the right drug is dispensed and administered, reducing errors and supporting compliance with hospital safety protocols.
+ Inventory Management – Automate stock counts, track lot numbers and expiration dates, and reduce waste caused by overstocking or expired medications.
+ Cold Chain Monitoring – Combine RFID with temperature monitoring to ensure vaccines and other sensitive products remain within required conditions during transport and storage.
+ Regulatory Compliance – Provide traceability for medications and vaccines, meeting FDA, EU, and other global standards for patient safety and supply chain oversight.
By automating these processes, RFID helps healthcare providers reduce risk, improve efficiency, and ensure that critical pharmaceuticals and vaccines remain safe and effective.